 It is the case that in a cubic 1:1 solid where one atom type is much larger than the other that the Cesium chloride type lattice is obtained, it can be thought of as a combination of basketballs and golf balls packed in a cubic manner with the golf balls in the gaps between the basketballs. The Cesium chloride structure is composed of interlocking simple cubic lattices of anions and cations. White cubic crystal hygroscopic density 3.99 g/cm3 melts at 645☌ vaporizes at 1297☌ very soluble in water, soluble in ethanol. It is a reagent in analytical chemistry, where it is used to identify ions by the color and morphology of the precipitate. Cancer radiation therapy, high pH therapy), in the diagnosis of myocardial infarction, and as a solute for ultracentrifugation.Ĭesium chloride is widely used medicine structure in isopycnic centrifugation for separating various types of DNA. Less than 20 tonnes of CsCl is produced annually worldwide, mostly from a caesium-bearing mineral pollucite.Ĭesium Chloride is a chemical reagent used in nuclear and radiologic medical treatments (e.g. Cesium chloride occurs naturally as impurities in carnallite (up to 0.002%), sylvite and kainite. CsCl changes to NaCl structure on heating. 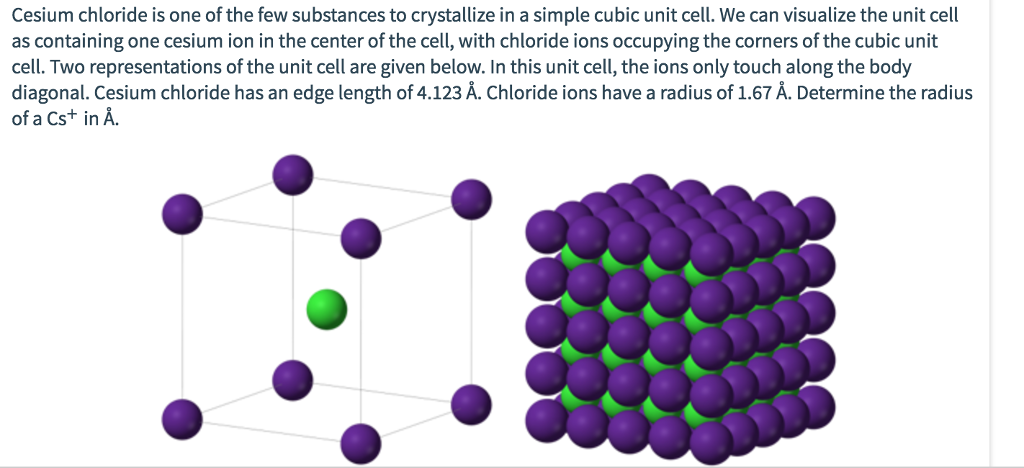
Its crystal structure forms a major structural type where each caesium ion is coordinated by 8 chlorine ions. 
This colorless solid is an important source of caesium ions in a variety of niche applications. Cesium chloride is the inorganic compound with the formula CsCl. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
